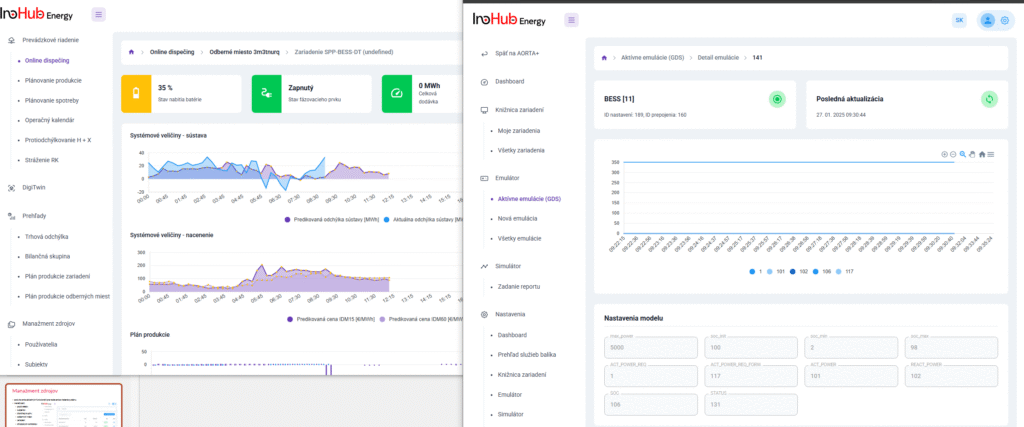
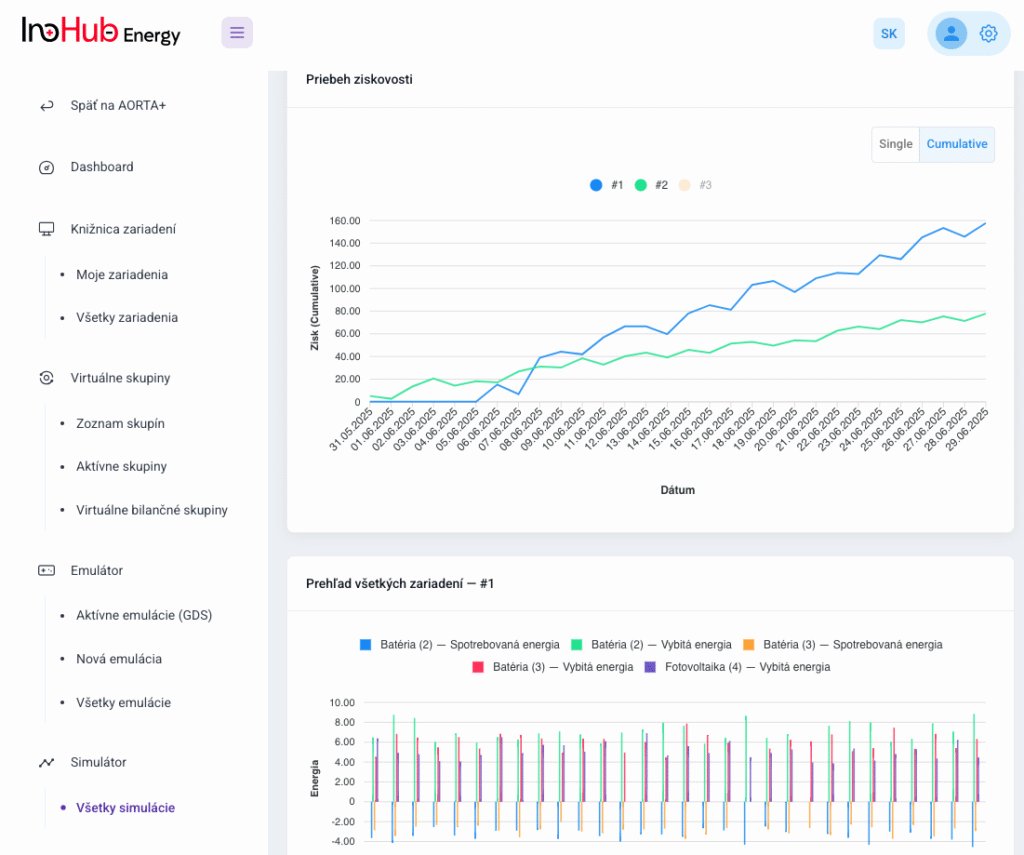
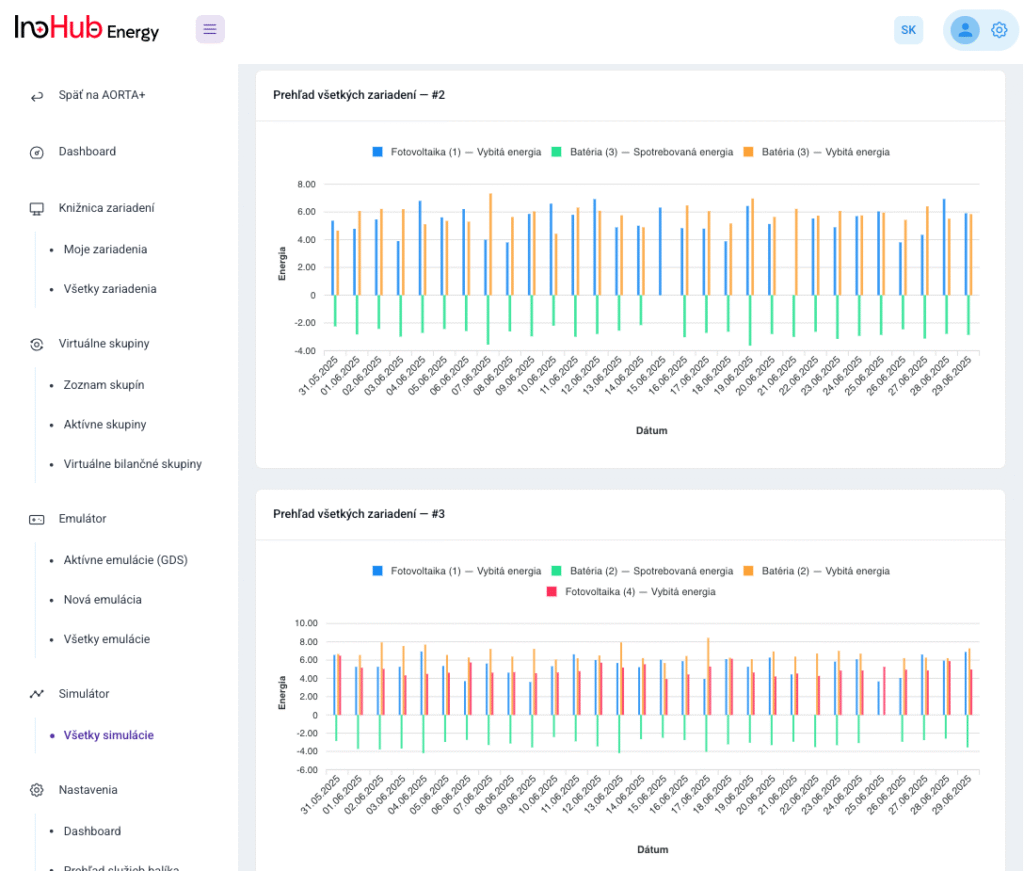
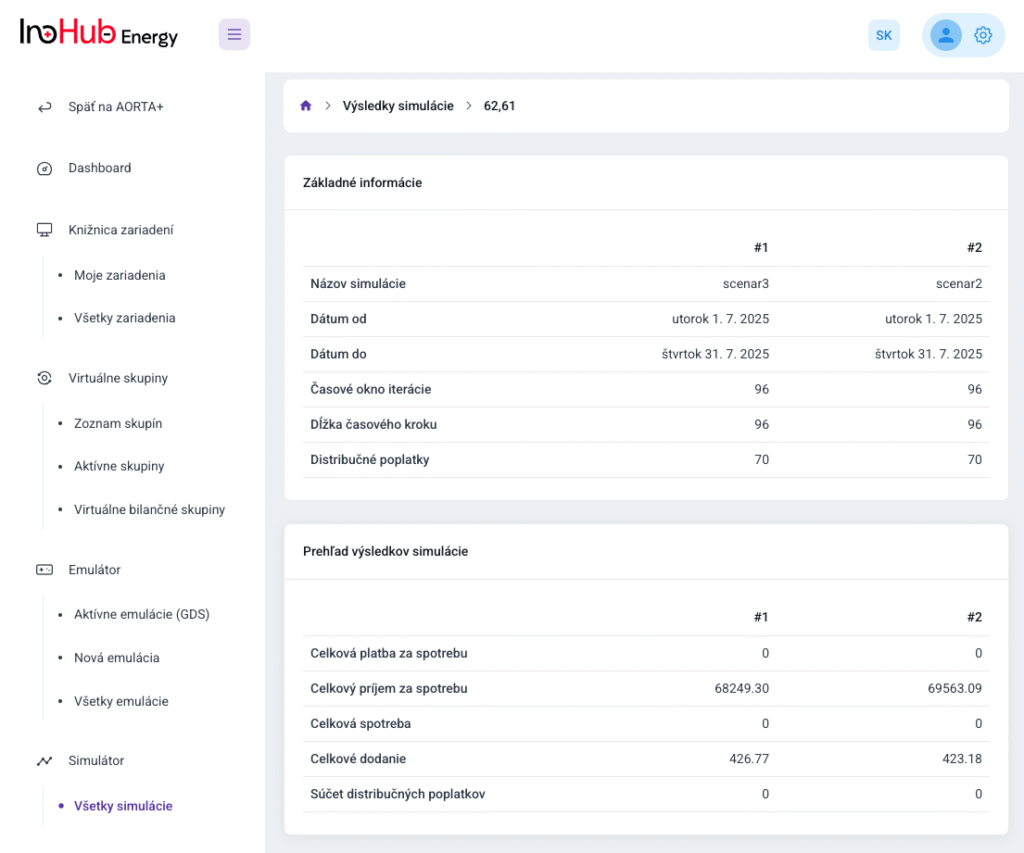
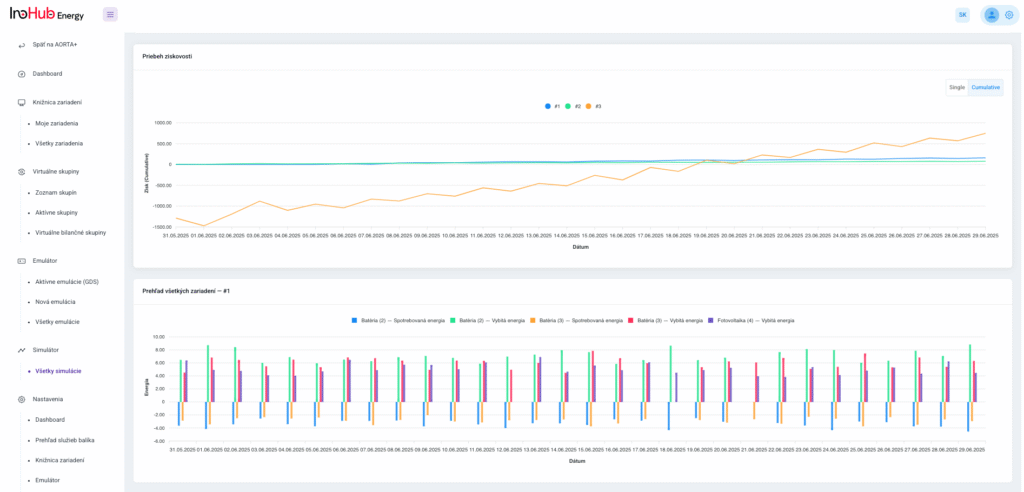
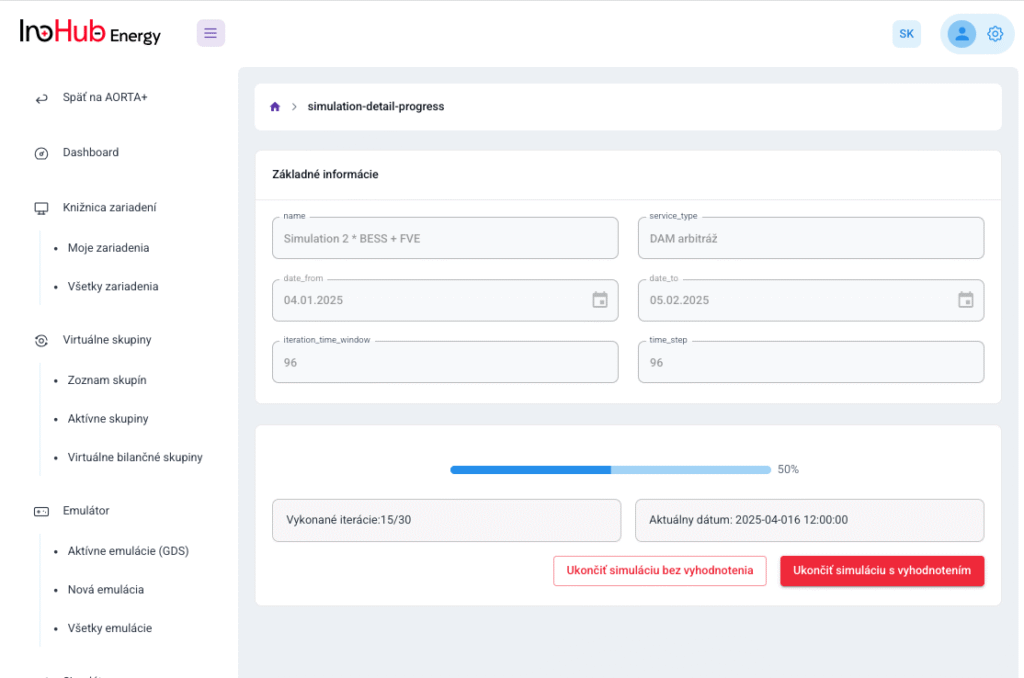
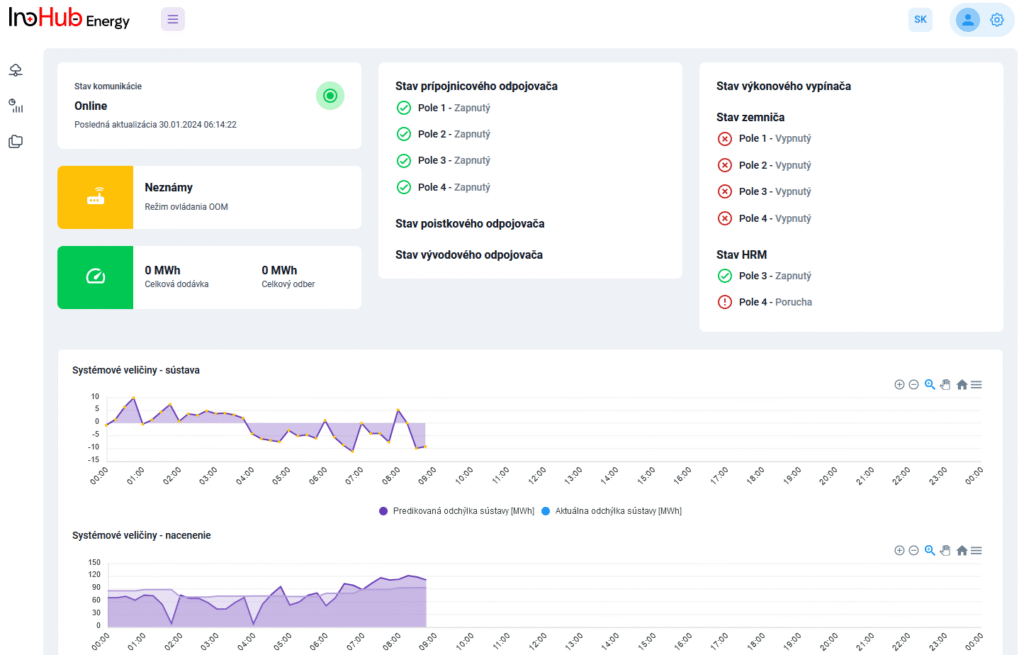
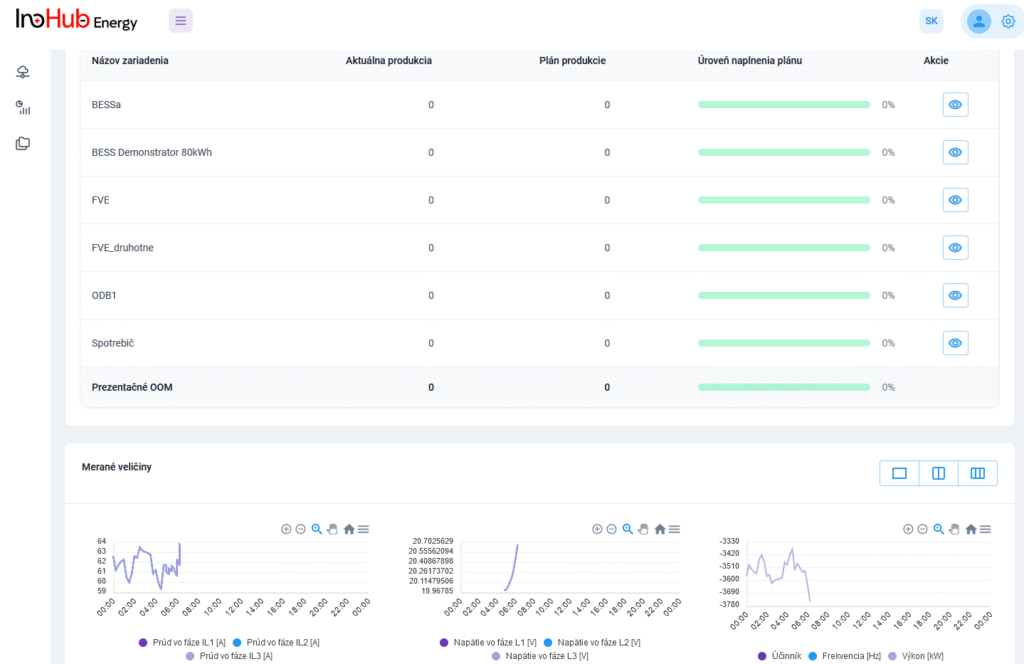
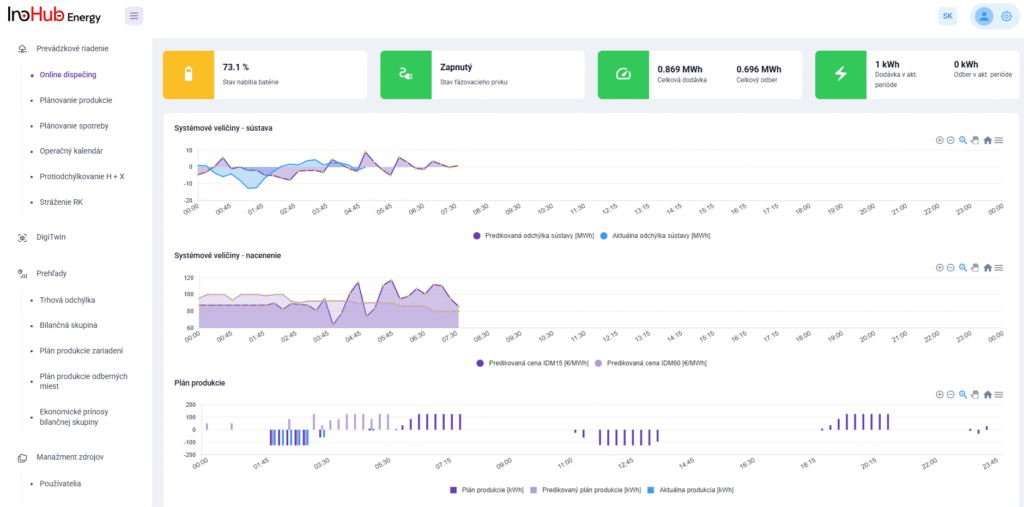
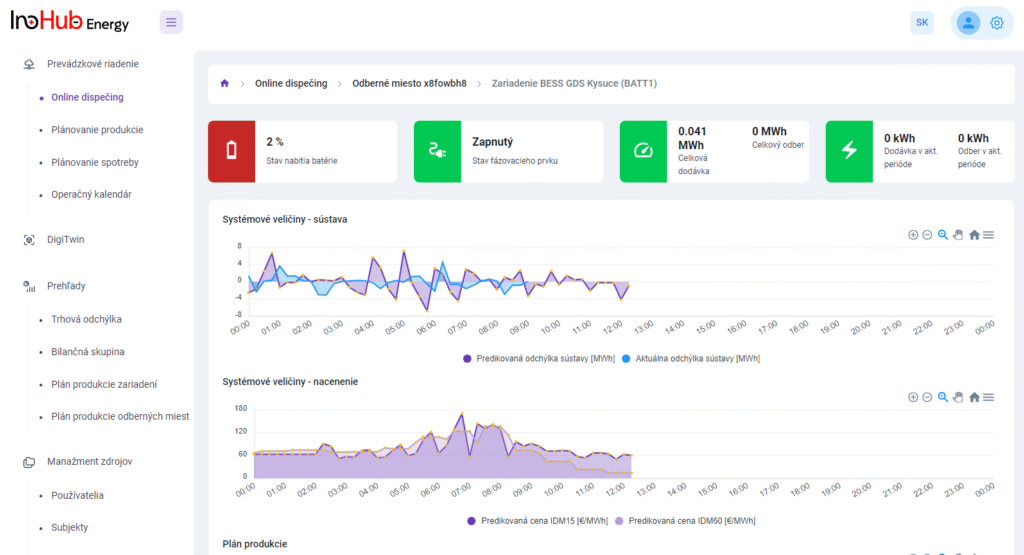
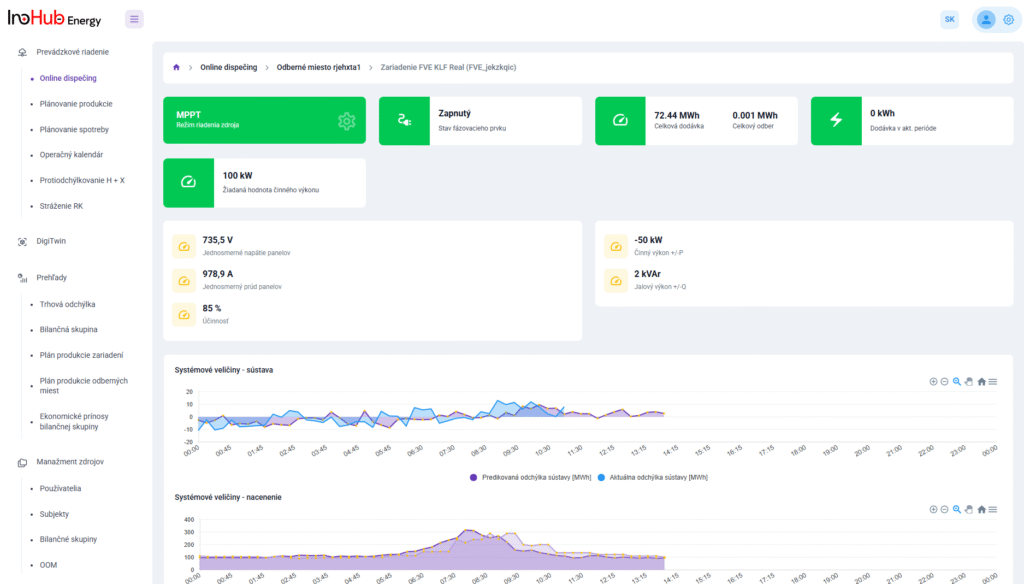
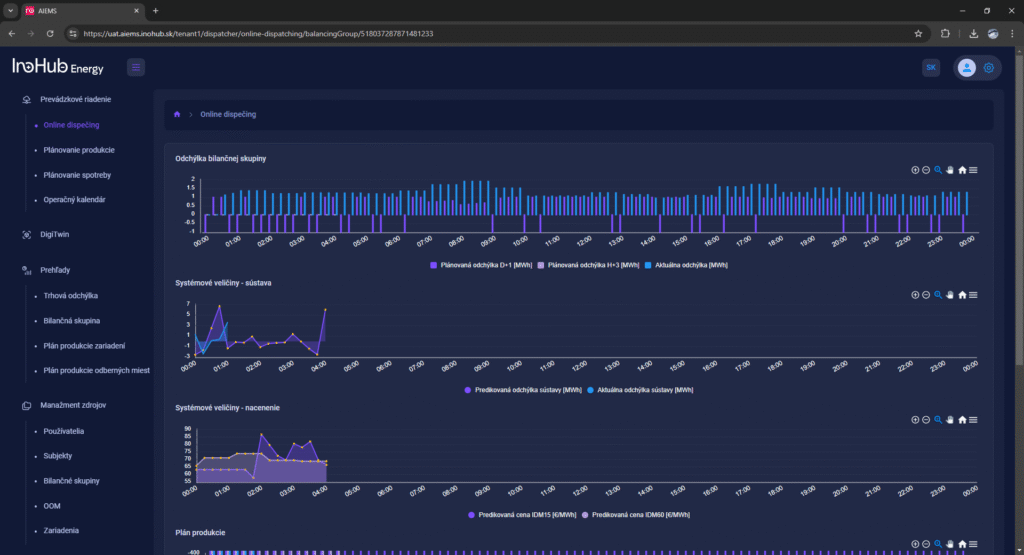
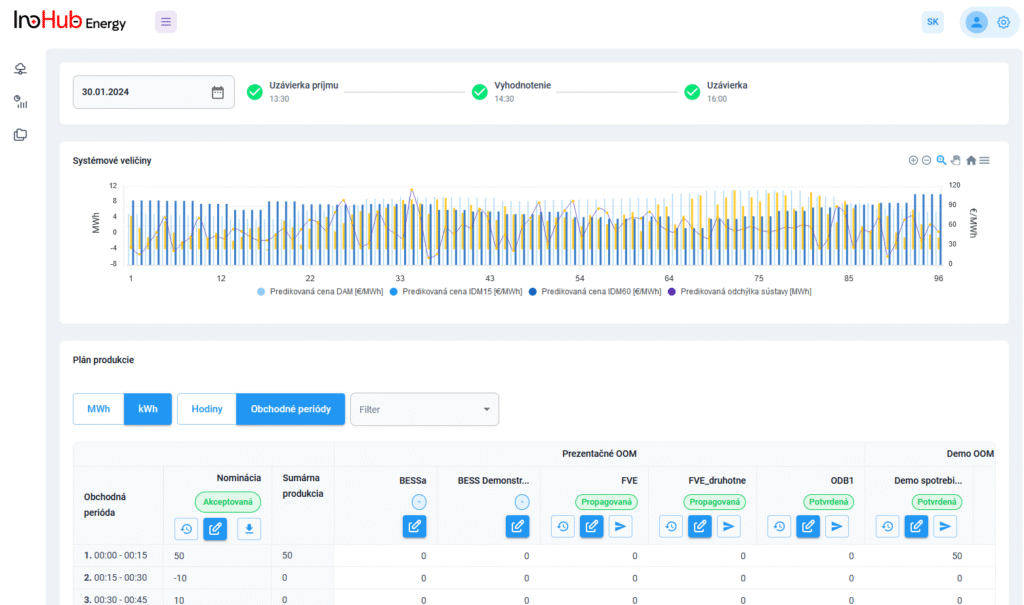
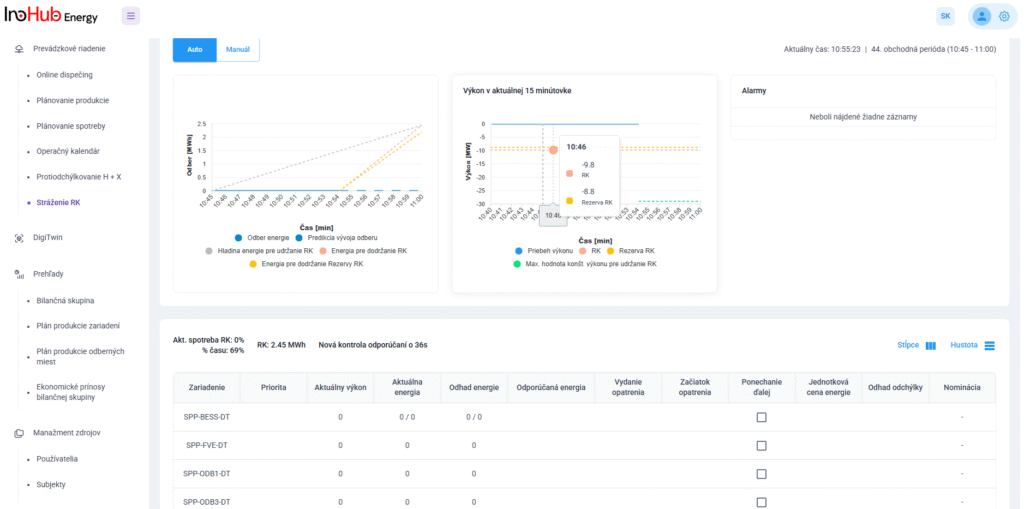
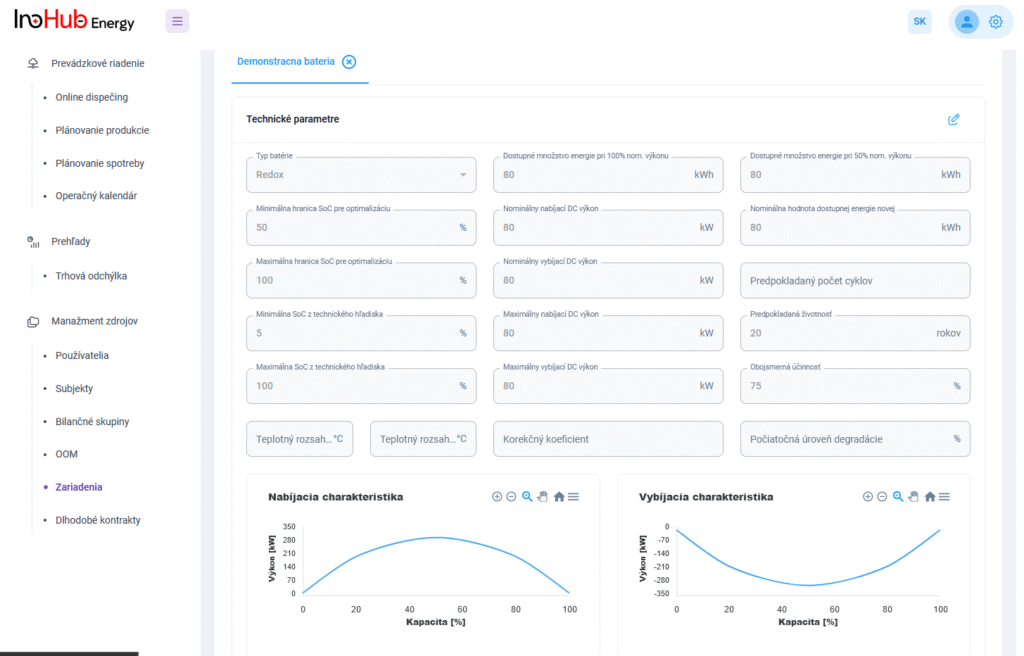
Among the different articles the importance of different components such as the bipolar plates, membranes, electrolytes on the battery performance has been depicted. But nothing has been reported about the place where the redox reactions take place, the electrodes. Commonly, carbonaceous electrodes are widely used in Redox Flow Batteries existing different alternatives such as carbon felts, carbon cloth and carbon papers. Different works have described the mechanism of the redox reactions of Vanadium Redox Flow Batteries. Even electrodes are carbonaceous materials, the redox reactions take place in the oxygen functional groups on the surface of the electrodes. The felt characteristics have significant effect on the battery performance, pressure drop, energy density and cost. According to InoHub stack design, carbon felts represent the most suitable candidate. As all the components of the complex redox flow batteries, carbon felts must be optimized to achieve better results. Different strategies have been employed in the literature to improve the poor wettability and electrochemical activity of carbonaceous electrodes:
- Thermal treatment: By treating the electrodes with high temperatures under different atmospheres the content of functional groups can be tuned.
- Gamma-ray irradiation to improve the mechanical properties of natural fiber-reinforced composites and fix their geometry.
- Use of composite materials: Different materials can be used to produce new electrodes promoting the electrochemical reactions on the electrode. Thess new materials usually present high oxygen-containing functional groups increasing the active sites in the electrodes.
- Electrochemical deposition: Carbon nanotubes and graphene oxide have been electrochemically deposited on the carbon felts increasing the active sites for the vanadium redox reactions. Usually, the electrochemical oxidation increases the oxygen-containing groups.
Among the different candidates and modifications studied in the literature, InoHub R&D team is studying the most promising strategy to optimize the performance of the electrodes using a cost-effective method. In this sense, the optimization of the electrodes will provide batteries with higher energy density and faster kinetics.
ACKNOWLEDGEMENT:
This work was supported by the project: IPCEI_IE_FLOW_BESS_012021